Tumor Lysis Syndrome Guidelines Nccn
Tumor lysis syndrome guidelines nccn. The National Comprehensive Cancer Network NCCN updated their guideline for chronic lymphocytic leukemia CLL to include key information on tumor lysis syndrome prophylaxis and treatment with duvelisib for disease with or without del 17pTP53 mutation. In addition to changes to the treatment regimen of CLLSLL the NCCN has made changes to the recommendations for the detection of tumor lysis syndrome TLS in this population. Monday August 31 2020 A case study published in JCO Oncology Practice reported a duvelisib treatment-emergent case of tumor-lysis syndrome in a patient with relapsed or refractory chronic lymphocytic leukemia CLL who had previously undergone chimeric antigen receptor CAR T-cell.
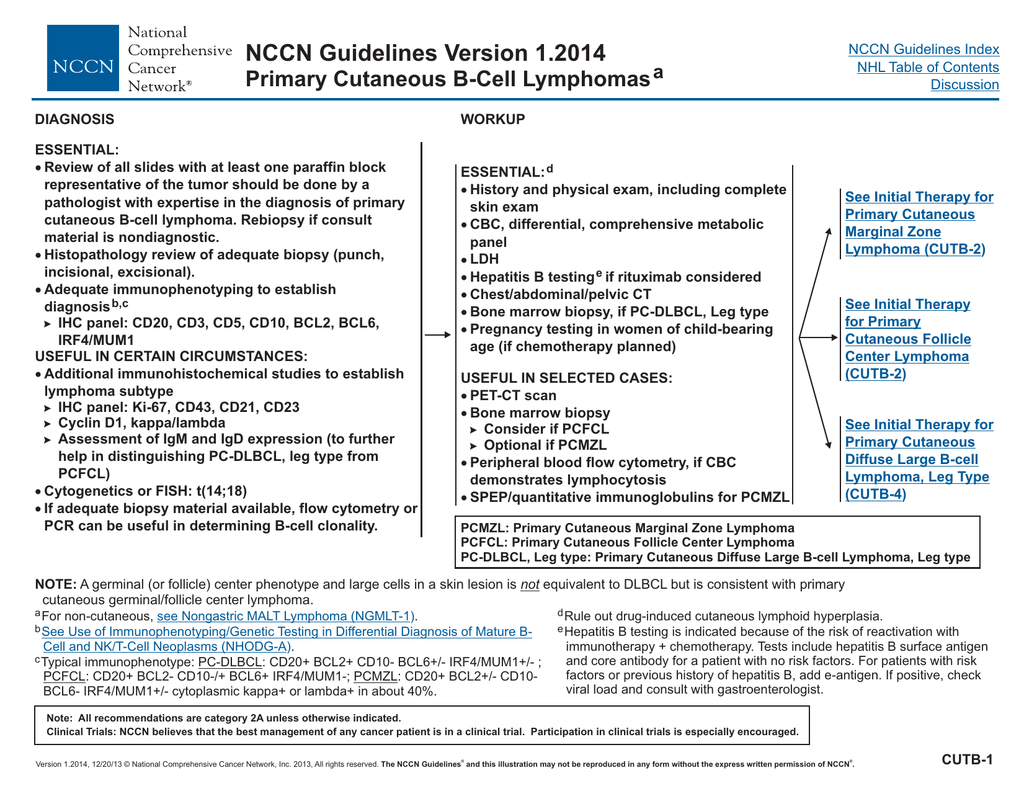
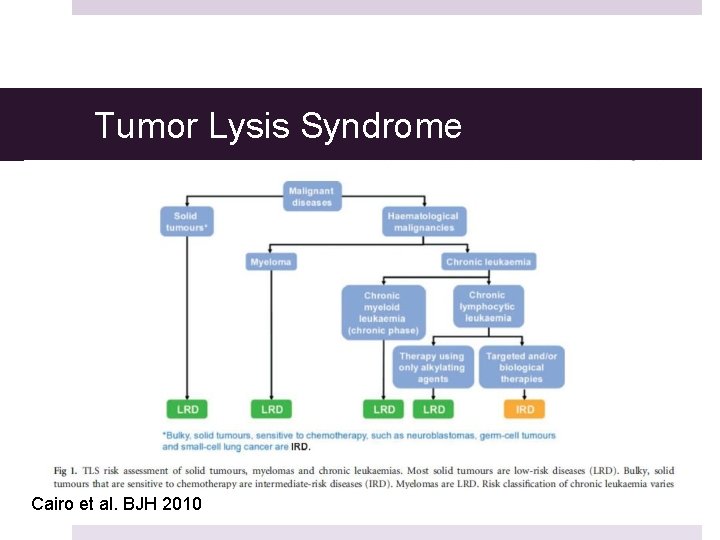
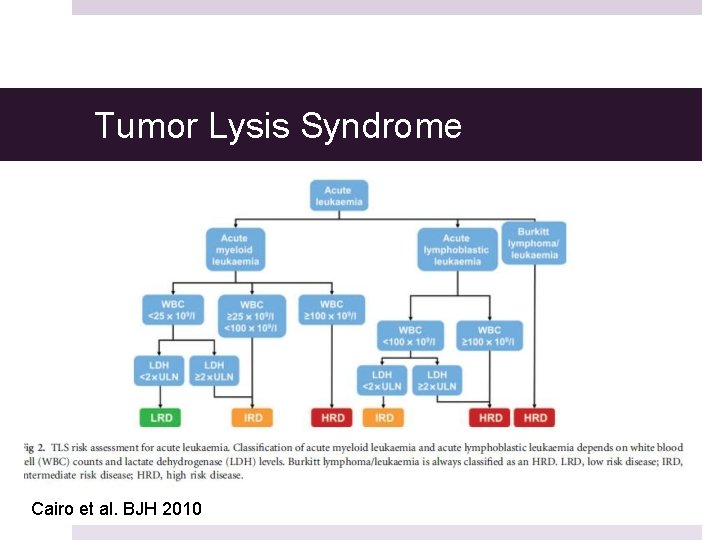
Zitella MS RN ACNP-BC AOCNUCSF Helen Diller Family Comprehensive Cancer CentersNCCN Medical Education Disclosure PolicyIt is NCCNs policy that every 12 months all faculty moderators activity planners and all internal planning staff participating in NCCN continuing education activities are. Standardized guidelines however are needed to aid in the stratification of patients according to risk and to establish prophylaxis and treatment recommendations for patients at risk or with established TLS. TLS identified by laboratory hallmarks of high potassium uric acid phosphorous lactate dehydrogenase and low calcium levels occurs when large amounts of tumors cells undergo lysis and their contents are released into.
Tumor lysis syndrome TLS has recently been subclassified into either laboratory TLS or clinical TLS and a grading system has been established. Tumor lysis syndrome guidelines nccn IMPORTANT SAFETY INFORMATION FOR CALQUENCE acalabrutinib capsules Severe and opportunistic infections Fatal and serious infections including opportunistic infections have occurred in patients with haematological malignancies treated with CALQUENCENCE. 3 and Table 2 in the Supplementary Appendix.
And for spontaneous TLS progressive disease after small-molecule inhibitor therapy and preexisting hyperuricemia NCCN 2020a. Nccn guidelines for tumor lysis syndrome Laura J. NCCN Updates Treatment Guideline for CLL.
Experts have issued management guidelines for the tumor lysis syndrome 2938 but further guidance awaits simple risk-prediction models that have a standardized definition of the tumor lysis syndrome and uniform supportive care guidelines for each cancer type. TLS is a potentially serious complication of anticancer therapy. TLS can be managed effectively if it is recognized early and preventative measures instituted prior.
Updates in the Management of Tumor Lysis Syndrome. A patient with 2 or more of the following abnormalities within 3 days before to 7 days after initiation of cancer treatment. This review summarizes current.
If left untreated its progression may cause acute kidney failure cardiac arrhythmias seizures loss of muscle control and death. Any clinician seeking to apply or consult the nccn content the nccn guidelines andor any derivative resources is expected to use independent medical judgment in the context of the individual clinical circumstances to determine any patients care or treatment.
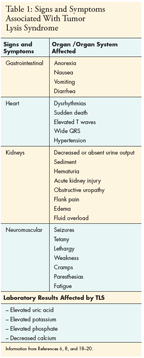
Clinical symptoms include hyperkalemia hyperphosphatemia hypocalcemia nausea vomiting diarrhea anorexia lethargy hematuria muscle cramps syncopy heart failure and cardiac.
3 and Table 2 in the Supplementary Appendix. Phosphate 65 mgdL children 45. The tumor lysis syndrome is the most common disease-related emergency in children and adults with hematologic cancers. TLS is a potentially serious complication of anticancer therapy. Any clinician seeking to apply or consult the nccn content the nccn guidelines andor any derivative resources is expected to use independent medical judgment in the context of the individual clinical circumstances to determine any patients care or treatment. And for spontaneous TLS progressive disease after small-molecule inhibitor therapy and preexisting hyperuricemia NCCN 2020a. Zitella MS RN ACNP-BC AOCNUCSF Helen Diller Family Comprehensive Cancer CentersNCCN Medical Education Disclosure PolicyIt is NCCNs policy that every 12 months all faculty moderators activity planners and all internal planning staff participating in NCCN continuing education activities are. The National Comprehensive Cancer Network NCCN updated their guideline for chronic lymphocytic leukemia CLL to include key information on tumor lysis syndrome prophylaxis and treatment with duvelisib for disease with or without del 17pTP53 mutation. Tumor lysis syndrome TLS has recently been subclassified into either laboratory TLS or clinical TLS and a grading system has been established.
Experts have issued management guidelines for the tumor lysis syndrome 2938 but further guidance awaits simple risk-prediction models that have a standardized definition of the tumor lysis syndrome and uniform supportive care guidelines for each cancer type. 3 and Table 2 in the Supplementary Appendix. Experts have issued management guidelines for the tumor lysis syndrome 2938 but further guidance awaits simple risk-prediction models that have a standardized definition of the tumor lysis syndrome and uniform supportive care guidelines for each cancer type. Uric acid 8 mgdL or 25 increase from baseline. Tumor lysis syndrome guidelines nccn IMPORTANT SAFETY INFORMATION FOR CALQUENCE acalabrutinib capsules Severe and opportunistic infections Fatal and serious infections including opportunistic infections have occurred in patients with haematological malignancies treated with CALQUENCENCE. Nccn guidelines for tumor lysis syndrome Laura J. Tumor lysis syndrome guidelines nccn By The ASCO Post Staff January 25 2020 The National Comprehensive Cancer Network NCCN has published new guidelines for treating children adolescents and young adults with pediatric aggressive mature B-cell lymphomas including Burkitt lymphoma and diffuse large B-cell lymphoma.






























Post a Comment for "Tumor Lysis Syndrome Guidelines Nccn"